 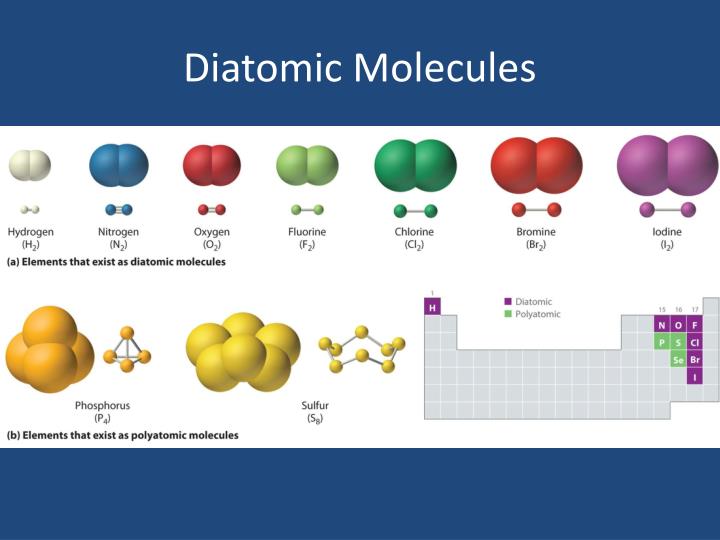
When drawing the number seven don’t neglect the seventh element, hydrogen. Shown in the case, should you take your finger and draw the number seven, beginning at nitrogen, you will see that six of the diatomic elements are arranged in the form of a number seven. If there’s ever a need to recognize these diatomic elements on the periodic table, they can be spotted by remembering the “seven rule” as well. 1 great way to remember that atoms form the blessed seven diatomic elements is to think of another mnemonic device: ”I Bring Cookies For Your New Home”. Instead, these molecules will always be paired together since they need to pool resources to possess enough electrons. 
That is, you’ll never find a nitrogen or fluorine atom, for instance, hanging out. However, with this greater temperature, they will also exist as gases.ĭiatomic elements are unique since the atoms which form them do not like to be alone. At room temperature, bromine and iodine commonly exist in the liquid kind. For example, the diatomic component oxygen has a formulation of O2, meaning that there are two different oxygen atoms present.Īt room temperature, you will find five diatomic elements, all of which exist at the gas type: hydrogen, nitrogen, oxygen, fluorine, and chlorine. If we notice the prefixed from the word diatomic, it comes from the Greek source of two.’ One big way to understand you are functioning with a diatomic element is to think about its own formulation.Įvery molecule has its own unique molecular formula, also for diatomic components, their formula regularly contains an addendum of 2, which performs two atoms in its arrangement. Each one of the elements in this noble group is gases. This oxygen, O2, and nitrogen, N2, have something in common: Both are known as diatomic elements.Īs pure components, diatomic elements are just seven specific molecules that are composed of two atoms. To be better precise, nitrogen constitutes 78% of the space whilst oxygen makes up 21%. There are major segments that form the planet’s air: oxygen, nitrogen, and even a tad sample of argon. Just how Many Diatomic Elements Are There?.Hydrogen has been proposed as an alternative fuel that is more environmentally friendly. Hydrogen is a diatomic element that is colorless and odorless gas at room temperature. A large percentage of our atmosphere is oxygen, and it plays a vital role in sustaining life on earth! Diatomic Element Hydrogen Oxygen is a diatomic element that is a gas at room temperature. Additionally, it is found in all living organisms. It is the most abundant element in our atmosphere. Nitrogen is a diatomic element that is a gas at room temperature. Iodine can be either a liquid or a solid depending on temperature, pressure, and other factors. Fluorine and chlorine are gasses at room temperature. The halogen elements have a single bond between the two atoms. Diatomic Halogen Elementsįour diatomic elements are halogens-Cl 2, F 2, Br 2, I 2. Some people also use the word ‘BrIClHOF’ (pronounced brinkle-hoff) which incorporates the chemical symbol of each element. ( Iodine, Hydrogen, Nitrogen, Chlorine, Bromine, Fluorine, Oxygen) ( Hydrogen, Nitrogen, Oxygen, Fluorine, Chlorine, Bromine, Iodine) He Needed Oxygen For Controlling Both Insects. ( Hydrogen, Nitrogen, Fluorine, Oxygen, Iodine, Chlorine, Bromine) Several example sayings are listed below or you can make up your own! This makes it easy to match the letter to the element. An intuitive order is to start with hydrogen, then go across the periodic table, and then down the halogens. They also form an upside-down ‘L’ shape.įinally, there are many sayings to remember the first letter of all the elements. 
All the elements are bordering on each other except for hydrogen in the standard periodic table layout. The halogen group also ends with ‘-gen’.Īnother technique is to memorize the shape of the elements on the periodic table (image below).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
